Confocal and Airyscan Fast
A confocal microscope is a high-performance fluorescence microscope that uses scanning optics to see only one very thin focal plane within the sample. Everything above and below this optical section is hidden, to give better resolution and a prettier picture. Thick samples can be imaged without physical cutting, and there are a myriad of vital stains available for labeling features of living samples. Model organisms can add their own fluorescent protein tags to a gene product, for precise labeling of specific cell components.
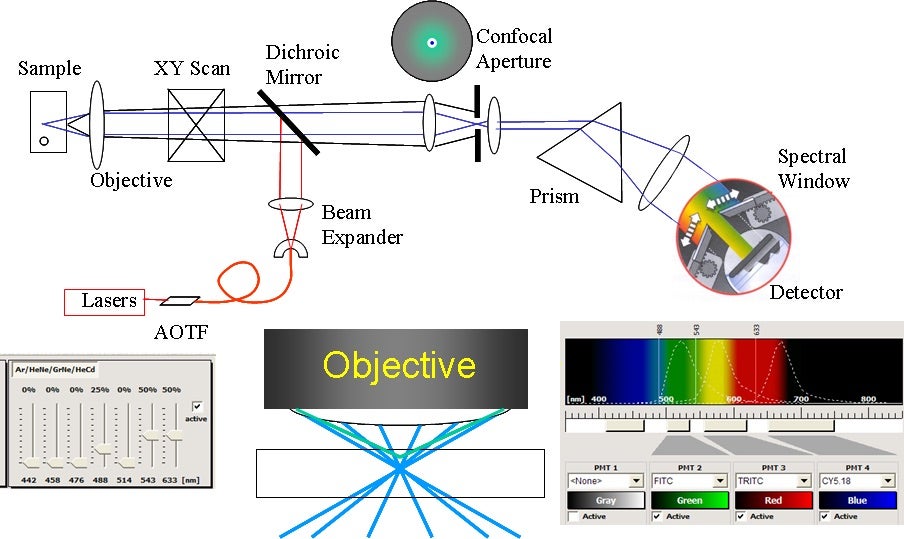
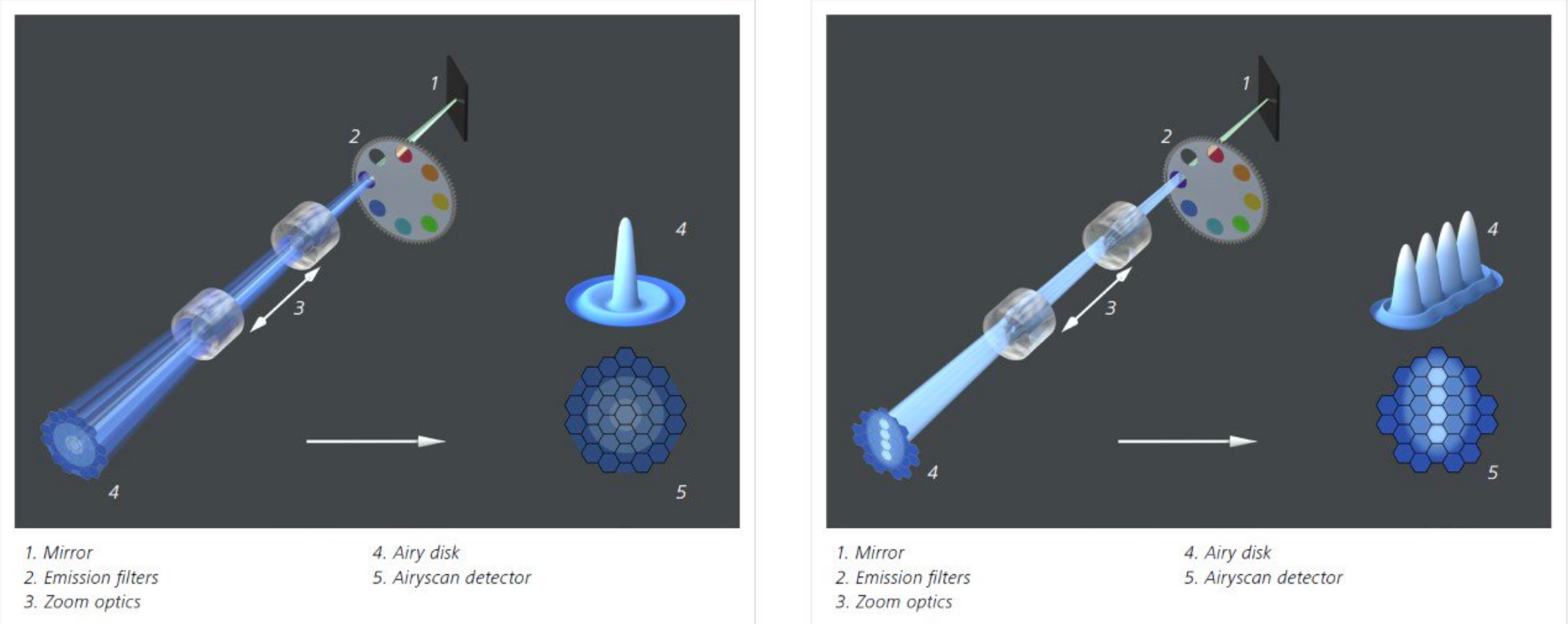
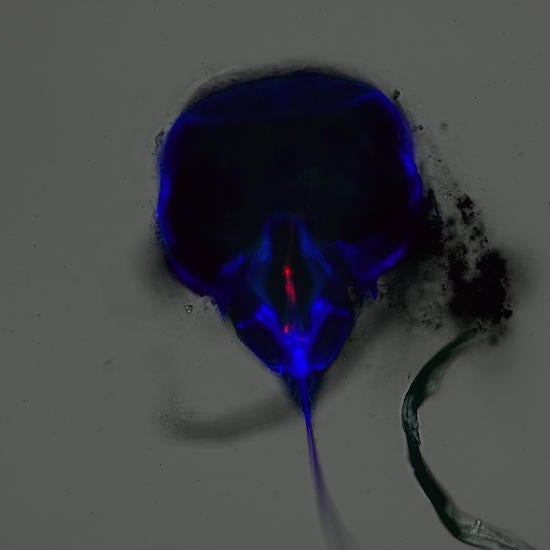
The two confocal technologies available in the core are spectral point scanning confocal (Leica or Zeiss) and Zeiss Airyscan Fast. Confocal scans a point of laser light into the sample, then returning signal is filtered through the confocal pinhole. The colors are spread across a set of detectors, which each take a chosen piece of the spectrum. This gives great flexibility, as any range of colors can be collected simultaneously. If multiple lasers are used, they can blink on and off rapidly, to prevent crosstalk between dyes and give cleaner colors. Spectral Unmixing can separate very similar dyes, or even remove autofluorescence to reveal dim underlying signal; such as in this whitefly head, coded blue, to reveal Alexa-647 labeled mosaic-virus inside the pharynx, where it waits to infect the next plant.
With Airyscan Fast, the laser light is stretched into a small line, which can sweep the sampled area many times faster. Returning light hits an array of pinholes, and the signal from each is compared to calculate a 50% sharper, noise-free image. The round laser can also be used with the Airyscan detector for a 70% sharpness improvement, but without the great increase in speed, or reduction in laser damage.
Our Keyence and Zeiss Inverted systems have an optional incubator stage inserts for long term experiments on mammalian cells, which can be kept at 37oC and 5% CO2 for extended time experiments.
Luminescence
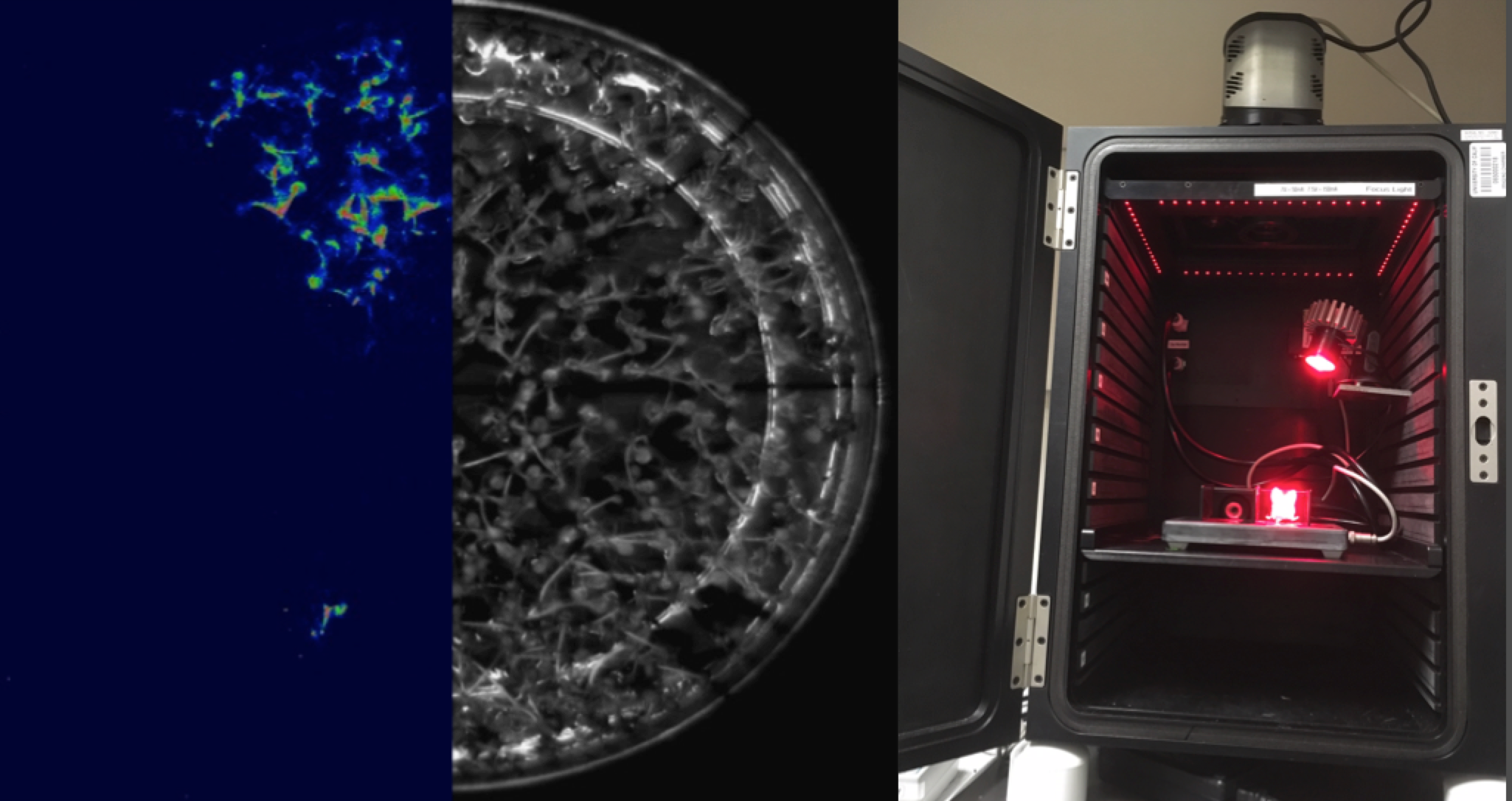
The detection limit of fluorescence technologies is set by background emission, which obscures very dim signals. But when Luciferase is exposed to its Luciferin substrate, it emits light in complete darkness. Our custom Luminescence Dark Box is an utterly dark enclosure, with a very sensitive cooled camera and bright lens. It can stare for up to 5 minutes, to locate extremely low copy number Luciferase expression on a macro scale. A mouse management manifold can keep pairs of mice comfortable for animal work. LED light engines have been added to allow quick fluorescence imaging in the same enclosure, using screw-on filters over the camera lens. A remote control piston has also been added, for example to tip a multiwell plate to mix in a reagent.
Scanning Electron Microscopy
Traditional SEM requires samples to be fixed, dried, and then gold coated, for imaging in a large electron column. Our new environmental SEM requires no preparation and can make publication quality images of fresh samples in a matter of minutes. It has optional voltage control, current control and vacuum control for greater versatility. At 5kV it can de-magnify to 10x; and at 20kV, the electrons have so much energy, they can make mineral samples glow (Cathode Luminescence) and reveal their age related growth rings. When the sample is loaded, an optical map is made of the whole stub, and very high pixel count images can be stitched together by drawing rectangles of interest on this color map.


3D Inspection & Measurement Microscope
New in May 2024, we have added the extremely versatile Keyence VHX-7000 3D inspection microscope for viewing samples as big as 4cmx4cm, which combines the long working distance advantage of a stereo scope with a multi-lens turret to offer a zoom range of 125x for macro and sub-micron micro data collection in a single instrument. Designed for industrial inspection, the extremely rugged instrument has calibrated telecentric lenses for exact measurement of distances, shapes, angles, areas etc, and can be tilted to view all sides of the sample. In three clicks, it produces extended-depth rubber-sheet renderings of the sample which can be spun around to any angle for better appreciation of 3D shape.
A pantograph hand micro manipulator can steady and gear-down hand movements for fine dissection work under the Keyence. Parts are on order to add better fluorescence capability to this instrument, which currently can use our NightSea set of LED light engines and magnetic clip-on filters to collect low power fluorescence images with the 20x-100x objective lens.
Fluorescence Stereo Microscope

A conventional Leica MZ-FL III stereoscope (Stereo1) with fluorescence and a 24 Megapixel color camera is also available. The camera is computer controlled for easier preview and adjustment of exposure time, bracketing, and ISO sensitivity. It has fluorescence filters for DAPI, CFP, GFP, YFP, dsRed and Cy5 type fluorophores, a ring light for top-down illumination, and a bottom-up illuminator with frosted or mirror modes for gentle or harsh or dark-field illumination. White light illuminaiton can be quickly shuttered on and off to facilitate screening of fluorescent v. non-fluorescent objects, such as seeds and nematodes.
.
Physical Sectioning
Sometimes a large sample needs to be cut carefully to expose an interior volume of interest. This can be done quickly on a cryostat. The sample is frozen in OCT mountant, then driven across a Tungsten Carbide blade, to cut sections up to 300µm thick. Alternatively, fresh tissue can be mounted in a water bath and sawn through with a vibrating razor or sapphire blade, on an oscillating tissue slicer.
An ultramicrotome is available for TEM sample preparation, for users already skilled in this work. Otherwise please consult the Central Facility for Advanced Microscopy and Microanalysis (CFAMM) in Bourns Hall, where the big electron microscopes are housed.
A high quality cryostat for cutting thin sections is housed within the School of Medicine Histology Core in Webber Hall.