Microscopy / Imaging Core
The Microscopy Core provides a comprehensive suite of confocal microscopes and supporting peripheral equipment. Under the supervision of a full-time imaging expert and Academic Coordinator, Dr. David Carter, the core has a range of optical imaging applications for use mammalian, insect, plant and other systems. There are three confocal microscopes: An inverted UV spectral system with incubator (Zeiss 880 Inverted Airyscan Fast); an upright visible system (Zeiss 880 Upright Airyscan Fast); and a user-friendly inverted confocal microscope (Leica SP5). We also have an all in one microscope for rapid non-confocal imaging, (Keyence BZ-X710), and a new upright 3D inspection microscope for superior white light imaging of 3D subjects up to 40x40mm and down to sub-micron (Keyence VHX-7000). Imaris Bitplane is used for 3D image rendering and analysis.
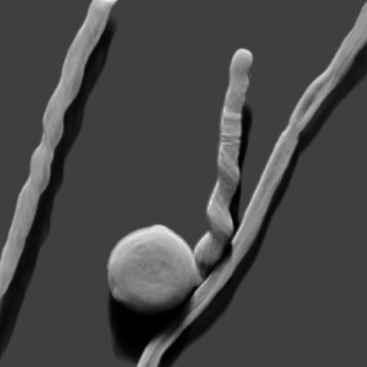
Other microscopy core equipment include a Hitachi TM4000PlusE II environmental SEM with EMS850 critical point dryer and Quorum SC7620 Gold/Palladium sputter coater; an ultramicrotome (Leica Ultracut T) with cryosectioning capability (Leica EM FCS); a fluorescence stereo microscope (Leica MZ FLIII), with a tethered 24 Megapixel Canon M50 camera for fluorescence and color imaging. There is a cryostat for rapid sectioning of frozen tissue (Hacker Bright); an oscillating tissue slicer (EMS5000); and a gene gun (BioRad PDS1000/HE Hepta). For macro imaging, there is a Nikon Multiphot enlarger stand, and a luminescence dark box (Stanford Photonics with Pixis 1024b camera) with 2-mouse manifold and LED light engines for fluorescence.
Which imager is best?
Since training is offered free of charge, it is a good idea to be trained on more than one system, so their differences are better understood. Generally, the Leica SP5 is the easiest to use, while the Zeiss 880s are faster, gentler, and higher resolution. Both brands have many laser colors to choose from, and complete flexibility over what colors are collected for confocal images.
Zeiss Airyscan is a new scanning method with 70% higher resolution; and Airy Fast is 50% sharper, but also extremely quick and gentle. These have more limited choice of color, as detection occurs through double-band filter wheels rather than via a spectral separator. But those filters are designed to work with the most common laser combinations, so multiple colors can be collected one after the other. The Zeiss 880 inverted has an incubator box for temperature control, to keep animal cells at 37°C and 5% CO2, and the Keyence BZ-X710 has a Tokei-Hit incubator insert option, along with many spare fluorescence cubes of many colors.
Leica has a very friendly user interface, including control knobs with the value displayed right above the knob. It has a clever laser delivery device which allows any combination of all laser lines to be launched at the sample. Its three PMTs and two HyD detectors can handle any practical mixture of fluorophores.
In many cases you do not need confocal and can get more done on the Keyence inverted BZ-X710. This robust inverted microscope can handle most sample types in fluorescence or bright field. It has easy navigation and can capture very large tiled images, multi-point time lapse, and video data. New in 2024 is an upright Keyence VHX-7000 inspection microscope, which has an incredible 125x zoom range. The extended depth of focus feature is especially well designed, for capturing the whole thickness of a sample in just three clicks, which can then be viewed from any angle to see its true 3d shape.